There’s more to the cell than its ‘powerhouse.’
By
Viviane Callier/ Knowable Magazine|
Released Nov 10, 2023 6:00 PM EST
This short article was initially included onKnowable Magazine
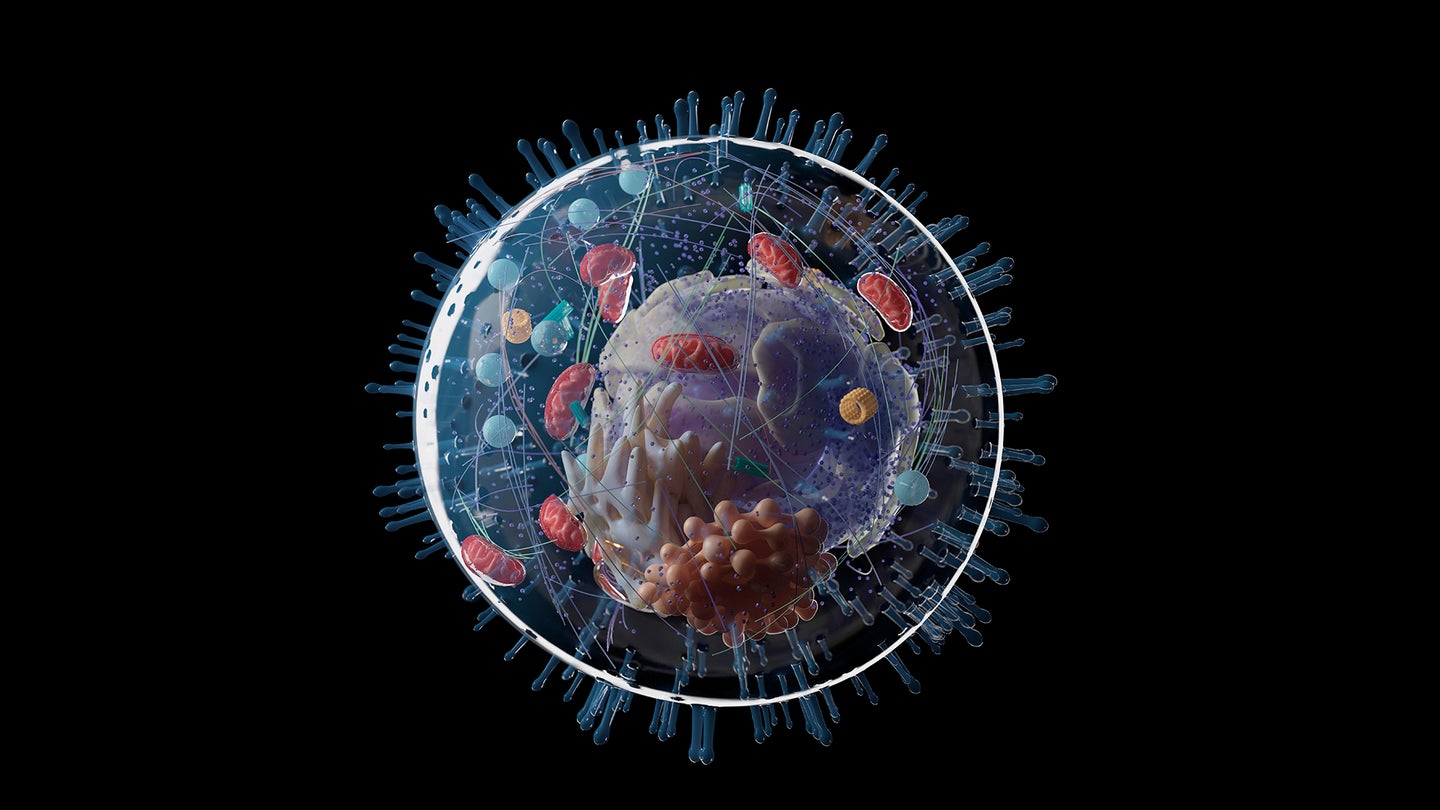
More than 1.5 billion years earlier, a memorable thing occurred: Two little, primitive cells turned into one. Maybe more than any occasion– disallowing the origin of life itself– this merger drastically altered the course of advancement on our world.
One cell wound up inside the other and progressed into a structure that schoolkids find out to describe as the “powerhouse of the cell”: the mitochondrion. This brand-new structure offered a remarkable energetic benefit to its host– a prerequisite for the later advancement of complex, multicellular life.
That’s just part of the story. Themitochondrionis not the just crucial structure within complex, eukaryotic cells. There’s the membrane-bound nucleus, safekeeper of thegenomeThere’s an entire system of internal membranes: the endoplasmic reticulum, the Golgi device, lysosomes, peroxisomes and vacuoles– vital for making, transferring and recycling proteins and other freight around the cell.
Where did all these structures originate from? With occasions lost in the deep past and couple of traces to act as evolutionary hints, it’s an extremely hard concern to deal with. Scientists have actually proposed numerous hypotheses, however it is just recently, with some brand-new tools and methods, that cell biologists have actually had the ability to examine the starts of this complex architecture and shed some light on its possible origins.
A microbial merger
The concept that eukaryotes stemmed from 2 cells combining go back more than 100 years however did not end up being accepted or popular till the 1960s, when the late evolutionary biologist Lynn Margulis articulated her theory of endosymbiosis. The mitochondrion, Margulis stated, most likely stemmed from a class of microorganisms called alphaproteobacteria, a varied group that today consists of the germs accountable for typhus and another one essential for the genetic modification of plants, amongst lots of others.
Absolutely nothing was learnt about the nature of the initial host cell. Researchers proposed that it currently was relatively made complex, with a range of membrane structures inside it. Such a cell would have can swallowing up and consuming things– a complex and energetically costly eukaryotic function called phagocytosis. That may be how the mitochondrion initially entered into the host.
This concept, called the “mitochondria late” hypothesis, does not discuss how or why the host cell had actually ended up being complex to start with.
In 2016, evolutionary biologistExpense Martincell biologistSven Gouldand bioinformaticianSriram Gargat the University of Dusseldorf in Germany, proposed a really various design referred to as the “mitochondria early” hypothesis. They argued that because no primitive cells today have any internal membrane structures, it appears really not likely that a cell would have had these over 1.5 billion years back.
Rather, the researchers reasoned, the endomembrane system– the entire mishmash of parts discovered inside complex cells today– might have developed not long after the alphaproteobacterium settled inside a reasonably basic host cell, of a kind from a class called archaea. The membrane structures would have occurredfrom bubbles, or blisters, launched by the mitochondrial forefather
Free-living germs shed blisters all the time, for all sorts of factors, Gould, Garg and Martin note, so it appears affordable to believe they ‘d continue to do that when confined inside a host.
Ultimately, these blisters would have ended up being specialized for the functions that membrane structures carry out today inside eukaryotic cells. They would even fuse with the host cell’s membrane, assisting to describe why the eukaryote plasma membrane consists of lipids with bacterial functions.
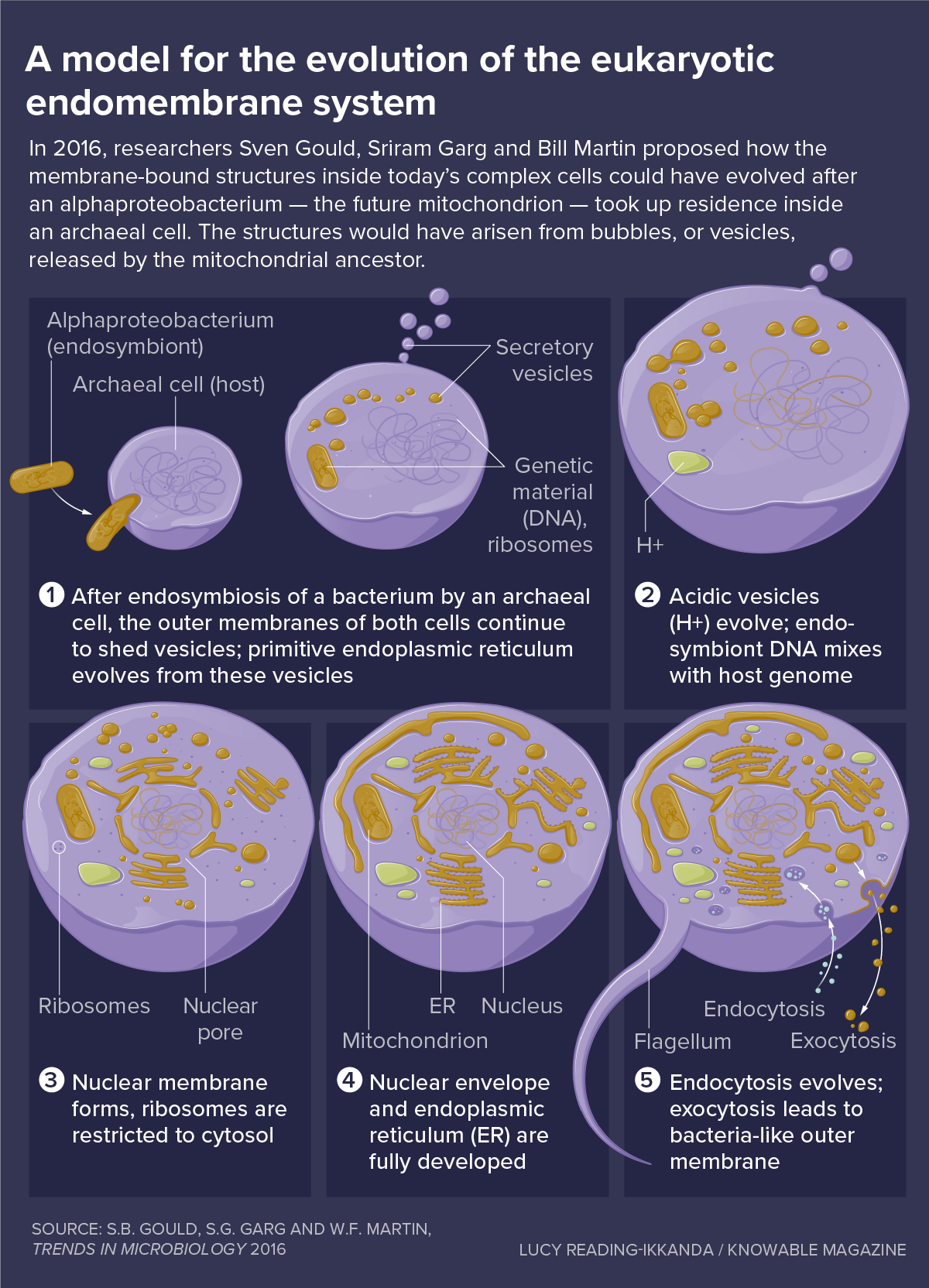
Blisters might have served an essential preliminary function, states biochemistDave Speijerof the University of Amsterdam. The brand-new endosymbiont would have produced lots of toxic chemicals called reactive oxygen types, by oxidizing fats and burning them for energy. “These ruin whatever, they are hazardous, specifically on the within a cell,” Speijer states. Sequestering them inside blisters would have assisted keep the cell safe from damage,he states
Another issue developed by the brand-new visitor might likewise have actually been assisted by making membranes barriers, Gould, Garg and Martin include. After the alphaproteobacterium showed up, littles its DNA would have blended with the genome of the archaeal host, disrupting crucial genes. Repairing this would imply progressing equipment to splice out these foreign pieces– today they’re referred to as introns– from themessenger RNA copies of genesso those protein-making directions would not be garbled.
That developed yet another issue. The protein-making equipment– the ribosome– works exceptionally quick, signing up with numerous amino acids together per second. On the other hand, the intron-removing system of the cell is sluggish, snipping out about one intron per minute. Unless the cell might keep the mRNA away from ribosomes till the mRNA was correctly processed, the cell would produce lots of ridiculous, ineffectiveproteins
The membrane surrounding the nucleus supplied a response. Acting as a spatial barrier, it enables mRNA splicing to end up in the nucleus before the intron-free mRNA is equated in the cell’s internal fluid, the cytosol. “This is the selective pressure behind the origin of the nucleus,” Martin states. To form it, blisters produced by the endosymbiont would have flattened and twisted around the genome, producing a barrier to keep ribosomes out however still permitting little particles to pass easily.
An inside-out description
Simply put, Gould, Garg and Martin’s hypothesis describeswhyendomembrane compartments developed: to fix issues developed by the brand-new visitor. It does not totally describe how the alphaproteobacterium got inside the host to start with, states cell biologistGautam Deyat EMBL in Heidelberg, Germany; it presumes the endosymbiont is currently within. “This is a huge issue,” Dey states.
An alternative concept, proposed in 2014 by cell biologistBuzz Baumof University College London (with whom Dey as soon as worked) and his cousin, University of Wisconsin evolutionary biologistDavid Baumis the”inside-outdesign. In this situation, the alphaproteobacterium and the archaeal cell predestined to be its ultimate host would have lived side by side for countless years in an intimate symbiosis, each depending upon the other’s metabolic items.
The archaeal cell would have had long protrusions, as seen on somemodern-day archaea that reside in close association with other microorganismsThe alphaproteobacterium would have nestled up versus these slim extensions.
Ultimately, the protrusions would have twisted around the alphaproteobacterium and confined it totally. Throughout the long stretch of time before that took place, the archaeal cell would have started some spatial department of labor: It would keep information-processing tasks in its center, where the genome was, while functions like protein structure would take location in the cytosol within the protrusions.

The power of the inside-out design, Buzz Baum states, is that it provides the cell eons of time, before the alphaproteobacterium ends up being totally confined, to progress methods to control the number and size of the mitochondrion and other membrane compartments that would ultimately end up being totally internal. “Until you can control them, you’re dead,” Buzz Baum states.
The design likewise discusses why the nucleus has the shape that it does; in specific, it offers a description for its uncommonly big pores. Seen from inside the center of an archaeal cell, the long protrusions would be openings that might naturally end up being huge pores like those, Baum states.
Essential, the inside-out design describes how the alphaproteobacterium would have entered the archaeal host in the very first location.
Still, the inside-out design has functions it requires to discuss. The mitochondrion would end up in the incorrect location– inside the endoplasmic reticulum, the network of tubes on which sit the cell’s protein-making ribosomes, as the archaeal protrusions covered around it. Therefore an extra action would be needed to get the alphaproteobacterium into the cytoplasm.
Martin’s primary objection is that the inside-out design does not supply an evolutionary pressure that would have triggered the nucleus or other membrane-bound compartments to emerge in the very first location. The inside-out design “is upside-down and in reverse,” Martin states.
The nucleus: A riddle in the center
The designs concur that the mitochondrion progressed from an alphaproteobacterium, they have extremely various concepts about the origin of the nucleus and other organelles.
In the Gould, Garg and Martin design, the source for all of the structures would have been blisters launched by the developing mitochondrion. Blisters to include reactive chemicals or cellular freight, and the capability to move this freight around, would have progressed really early. The nucleus would have come later on.
In the inside-out design, the nucleus was, basically, the remains of the archaeal cell after it covered its membranes around the alphaproteobacterium. It would have appeared right away. The endoplasmic reticulum likewise would have formed early, developed from those squished-together protrusions. Other organelles would have come later on– emerging, Buzz Baum states, from buds of archaeal membrane.
Hence the designs likewise alter forecasts about the chemical nature of the membranes of cell organelles– a minimum of initially– and how today’s complex cells came to have membrane lipids that are all chemically like the ones in germs, not archaea.
In the Gould, Garg and Martin design, in the starting all the membranes other than for the host cell’s outer one would have been bacterial, like the membranes of the brand-new citizen. As bacterial blisters merged with this archaeal external membrane, the bacterial lipids would gradually change the archaeal ones.
In the inside-out design, the membranes of the nucleus and endoplasmic reticulum– and most likely others– would have been archaeal, like the host, to begin. Just in the future, after genes from the bacterial genome moved over to the archaeal genome, would the lipids end up being bacterial in nature, Baum recommends.
How to evaluate these concepts? Through experiments, cell biologists are beginning to peek methods which easy blisters might have diversified into various organelles with unique tasks– by handling various shapes, like the layered membrane stacks of the contemporary endoplasmic reticulum or the Golgi body, or by winding up with various proteins inside them or on their membranes.
They are likewise highlighting the dynamism of the modern-day mitochondrion– and its prospective to generate brand-new membrane structures.
Take, for instance, the compartment that Speijer believes progressed early in order to handle reactive oxygen types: the peroxisome.
In 2017, cell biologistHeidi McBrideof McGill University in Montreal reported that cells doing not have peroxisomes mightcreate them from scratchDealing with mutant human fibroblast cells without peroxisomes, her group discovered that these cells put proteins that are necessary for peroxisome function into mitochondria rather. The mitochondrial membrane launched them as little bubbles, or blisters.
These blisters, or proto-peroxisomes, grew into real peroxisomes when they merged with another kind of blister originated from endoplasmic reticulum, which bring a 3rd needed peroxisome protein. “It’s a hybrid organelle,” McBride states.
For McBride, this is a sign that peroxisomes– and most likely other organelles– initially originated from mitochondria (not specifically from the endoplasmic reticulum, as formerly thought). “The existence of mitochondria introduced the biogenesis of brand-new organelles,” she states. “In the case of peroxisomes, it’s rather direct.”
Other mitochondrion shenanigans have actually likewise been kept in mind.
a 2021 reportfrom the laboratory of biochemistAdam Hughesat the University of Utah discovered that when yeast cells are fed poisonous quantities of amino acids, their mitochondria will shed blisters that are packed with transporter particles. The transporters move amino acids into the blisters, where they will not toxin the mitochondria.
Hughes likewise found that the blisters shed by the mitochondria can formlong, tubule-like extensions with several layerssimilar to the layered stacks of the endoplasmic reticulum and the Golgi body. The structures continue the cell for a long period of time. “They’re certainly their own special structure,” Hughes states.
And in 2022, immunologistLena Pernasnow at UCLA, revealed that multilayered, mitochondria-derived structures can form in other contexts, too. When a cell is contaminated by the parasiteToxoplasmaher group discovered, themitochondria surround the parasite and modification shapeThe parasite reacts, and the result is that the mitochondrion winds up shedding big littles external membrane.
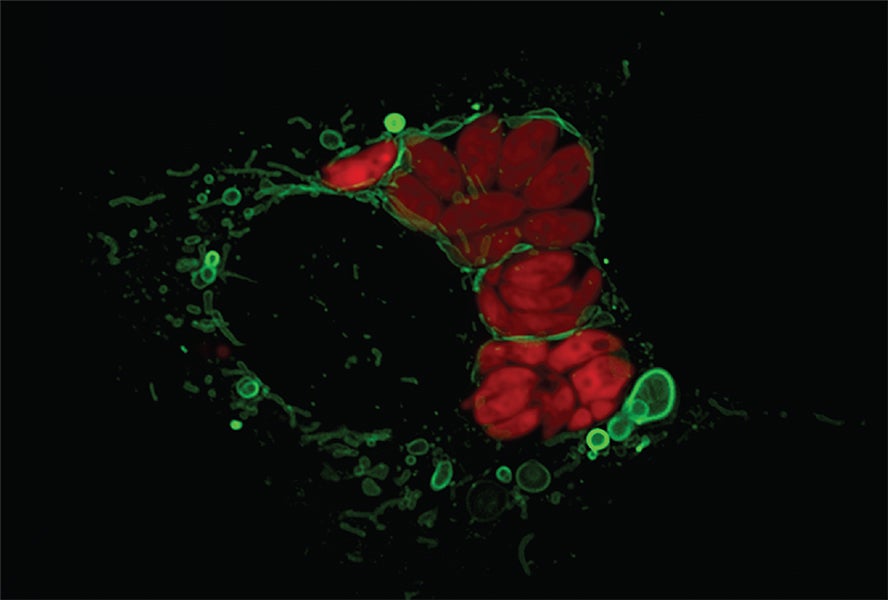
Pernas, who discussedmitochondrial improvementin theYearly Review of Physiologyin 2016, just recently found that these structures, which at first appear like basic blisters, likewise can grow and handle more intricate shapes, such as stacks of sheet-like layers. What’s more, the tension of infection modifications what sorts of proteins are put on these shed little bits of mitochondrial membrane. Such modifications unlock for the stacked sheets to act in various methods than they typically would, providing the chance to handle brand-new tasks, Pernas states.
The more Pernas and Hughes study these structures– discovered in rather various cells and conditions– the more comparable they look. It’s alluring, states Hughes, to envision how a structure like this, forming in the early days of eukaryote development, might have developed over eons of natural choice into a few of the endomembrane compartments existing in cells today.
It might never ever be possible to understand for sure what took place such a long time back. By exploring what can occur in today’s living bacterial, archaeal and eukaryotic cells, researchers can get more clearness on what was possible– and even likely. A cell moves into another cell, bringing advantages however likewise issues, triggering a complex waterfall. And after that, McBride states, “all this things blossoms and blooms.”
This post initially appeared inKnowable Magazinean independent journalistic undertaking from Annual Reviews. Register for thenewsletter
Discover more from CaveNews Times
Subscribe to get the latest posts sent to your email.

























![Exploring the Serene Beauty of Nature: A Reflection on [YouTube video title]](https://cavemangardens.art/storage/2024/04/114803-exploring-the-serene-beauty-of-nature-a-reflection-on-youtube-video-title-360x180.jpg)























